Systemic Lupus Erythematosus, SLE, is one of the more complex conditions in veterinary medicine, not because any single aspect of it is impossible to understand, but because it affects so many systems simultaneously and presents differently from dog to dog. It is a chronic autoimmune disease in which the immune system loses its ability to distinguish between the body’s own cells and foreign threats, and begins attacking healthy tissue across multiple organs. Joints, skin, kidneys, blood cells, and neurological tissue can all be involved, sometimes together, sometimes in shifting combinations. Early diagnosis matters significantly: the sooner SLE is identified and managed, the better the chance of controlling it before serious organ damage accumulates.
What is Systemic Lupus Erythematosus in Dogs?
In a healthy immune system, the body’s defences are targeted: they attack pathogens and abnormal cells while leaving healthy tissue alone. In autoimmune diseases, this targeting breaks down. SLE is characterised by the production of autoantibodies, antibodies directed against the body’s own nuclear material (DNA and proteins within cell nuclei), which form immune complexes that deposit in tissues throughout the body, triggering inflammation wherever they accumulate.
What distinguishes SLE from localised autoimmune conditions is its systemic nature. A dog with autoimmune skin disease has immune dysregulation confined to one system. A dog with SLE has immune dysregulation that can affect the kidneys, joints, blood cells, and skin simultaneously, or sequentially as the disease waxes and wanes. This makes SLE both harder to diagnose and more demanding to manage than single-organ autoimmune disorders. A broader context of autoimmune disease in dogs provides a detailed overview of the disease category.
Symptoms of Systemic Lupus Erythematosus in Dogs
SLE is sometimes called “the great imitator” in veterinary medicine because its symptoms overlap with many other conditions and fluctuate in intensity, improving during remissions and worsening during flares. No single symptom is pathognomonic for SLE; the diagnosis is made by recognising a pattern across multiple systems.
Common Clinical Signs
- Fever – often recurrent, of unknown origin, resistant to antibiotics (because it is immune-mediated rather than infectious)
- Joint pain and lameness – polyarthritis is one of the most consistent presentations; joints are swollen, warm, and painful, and the lameness may shift from one limb to another as different joints flare
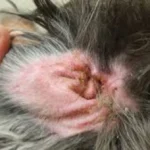
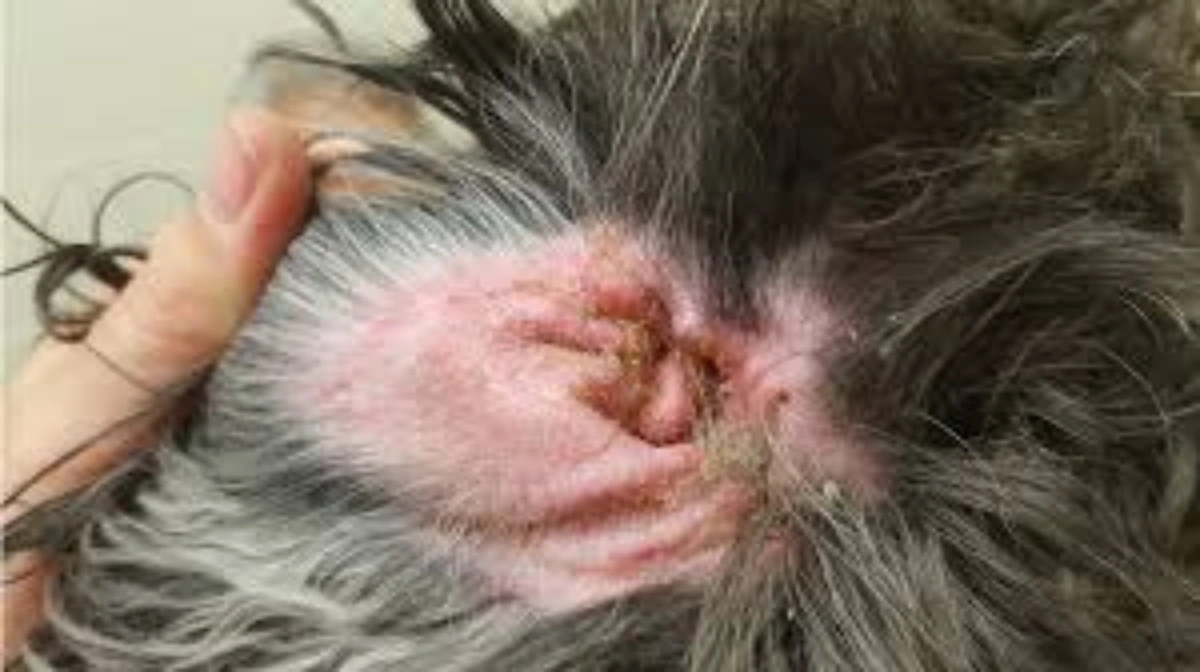
- Skin lesions – depigmentation of the nasal planum, crusting and ulceration around the face and muzzle, scaling and hair loss across the body; sun exposure can exacerbate cutaneous involvement
- Hair loss (alopecia) – diffuse or localised, often associated with the skin lesions
- Mucosal ulcers – oral ulceration is seen in some cases
- Kidney involvement (lupus nephritis) – proteinuria, increased thirst and urination, and eventually azotaemia as renal function declines; renal disease is one of the most prognostically significant manifestations of SLE
- Haematological abnormalities – immune-mediated haemolytic anaemia (IMHA), thrombocytopenia, or leukopenia may occur as part of SLE; pale gums, bruising, or petechiae may reflect these concurrent blood disorders
- Swollen lymph nodes – generalised lymphadenopathy reflecting systemic immune activation
- Lethargy and general malaise – persistent and disproportionate to any obvious single cause
- Neurological signs – less common, but seizures, behavioural changes, or peripheral neuropathy can occur in SLE cases with CNS involvement
The waxing-and-waning nature of SLE means a dog may appear to improve temporarily without treatment, which can delay diagnosis. The pattern over time – multiple systems affected, symptoms that recur despite apparently appropriate treatment, is the diagnostic key.
Causes of Systemic Lupus Erythematosus in Dogs
SLE does not have a single identifiable cause. Like most autoimmune conditions, it is understood as the product of genetic predisposition interacting with environmental or physiological triggers that tip the immune system into dysregulation.
Genetic predisposition – certain breeds are overrepresented in SLE case literature: German Shepherds, Beagles, Irish Setters, Shetland Sheepdogs, Afghan Hounds, and Collies. This does not mean other breeds are immune; it reflects lines with inherited tendencies toward immune dysregulation.
Sex and hormonal factors – female dogs appear to develop SLE at higher rates than males, mirroring the human pattern, suggesting hormonal influences on immune regulation may play a role.
SLE is not contagious – it cannot be transmitted between dogs or from dogs to people. It is an internally driven immune dysfunction, not an infectious process.
Risk Factors and Triggers
The exact triggers that initiate SLE in a genetically predisposed dog are not always identifiable, but several have been associated with disease onset or flares:
- Ultraviolet light exposure – sunlight is a documented trigger for cutaneous flares and possibly systemic exacerbation; this is particularly relevant for dogs with depigmented or lightly pigmented skin who are regularly outdoors
- Certain medications – drug-induced lupus-like syndrome has been reported in dogs, associated with some antibiotics, anticonvulsants, and other agents; a history of recent medication changes is an important part of the clinical workup
- Infections – viral or bacterial infections may trigger immune activation that precipitates an autoimmune response in susceptible individuals
- Stress – physical or psychological stress is recognised as a potential flare trigger in established SLE cases
- Hormonal changes – oestrus, pregnancy, or underlying hormonal conditions may influence immune activity
More resources on the range of conditions that can follow immune dysregulation are available on the VOSD blog.
Related Videos
 ▶
▶ ▶
▶ ▶
▶Diagnosing Systemic Lupus Erythematosus in Dogs
SLE diagnosis is notoriously challenging because no single test confirms it, and the symptoms overlap with many other conditions. Most published diagnostic criteria require a dog to meet a minimum number of clinical and laboratory criteria simultaneously.
Antinuclear antibody (ANA) test – the most diagnostically significant single test for SLE. Detects autoantibodies directed against nuclear material. A positive ANA in a dog with compatible multi-system clinical signs is strong evidence for SLE. However, ANA can occasionally be positive in other conditions, and a negative ANA does not definitively exclude SLE in early or mild disease.
Complete Blood Count (CBC) – identifies anaemia, thrombocytopenia, or leukopenia; the haematological changes may be direct manifestations of SLE or reflect concurrent immune-mediated blood disorders.
Urinalysis and urine protein: creatinine ratio (UPC) – assesses renal involvement; proteinuria is a key indicator of lupus nephritis, which can be present before overt kidney disease signs are detectable.
Serum biochemistry panel – evaluates renal function (creatinine, BUN), liver enzymes, and albumin; provides a baseline for monitoring organ function over time.
Joint fluid analysis – in dogs with polyarthritis, arthrocentesis and cytology of synovial fluid typically reveals a non-septic inflammatory pattern (high WBC count without infectious organisms), consistent with immune-mediated joint disease.
Skin biopsy – in dogs with significant dermatological involvement, skin biopsy with immunofluorescence staining can show characteristic immune complex deposition at the dermal-epidermal junction (the “lupus band”).
Coombs test – confirms immune-mediated haemolysis if IMHA is present alongside other SLE manifestations.
Imaging – joint radiographs and abdominal ultrasound are used to assess joint changes and organ involvement (spleen, lymph nodes, kidneys).
The diagnostic process for SLE is typically one of systematically excluding other conditions while accumulating evidence from multiple abnormalities pointing toward autoimmune multi-system disease.
Prognosis and Treatment Options
SLE is a chronic condition; it is managed over time rather than cured. Prognosis depends heavily on which organ systems are involved and how they respond to treatment. Dogs with predominantly joint and skin involvement tend to have better outcomes than those with significant renal disease, where long-term prognosis is more guarded, as kidney function can continue to decline despite treatment.
Corticosteroids – prednisolone at immunosuppressive doses is the foundation of initial treatment; effective at suppressing immune activation and reducing inflammation across all affected systems. Side effects with prolonged high-dose use (increased thirst and urination, weight gain, increased infection susceptibility) require monitoring and dose reduction to the lowest effective maintenance level.
Second-line immunosuppressants – azathioprine, cyclosporine, mycophenolate, or leflunomide are added when corticosteroids alone are insufficient, when side effects limit their use at therapeutic doses, or when the disease is refractory. These agents require more intensive monitoring for toxicity.
Hydroxychloroquine – an antimalarial agent used in human SLE, increasingly explored in veterinary medicine for dogs with refractory or steroid-dependent disease; limited but growing evidence base in dogs.
Management of organ-specific complications – lupus nephritis requires additional renal-protective measures, including ACE inhibitors, dietary protein adjustment, and blood pressure management; haematological complications (IMHA, thrombocytopenia) are managed according to their specific protocols.
Sun avoidance – practical and meaningful for dogs with cutaneous involvement; limiting midday outdoor activity and avoiding prolonged direct sun exposure reduces flare frequency in affected dogs.
Long-Term Management and Monitoring
SLE is a lifelong condition for most dogs. The management framework includes:
- Regular CBC, biochemistry, and urinalysis monitoring – typically every 1–3 months during active disease or treatment adjustment, extending to quarterly or biannual once stable
- Tracking ANA titres as one indicator of disease activity, though clinical signs and organ function tests are weighted more heavily
- Gradual tapering of immunosuppressants during remission, watching closely for clinical relapse
- Documenting flare patterns and identifying individual triggers where possible
- Owner education on recognising early relapse signs – returned joint pain, fever, skin changes – so treatment can be adjusted before the flare becomes severe
Preventing Complications in Dogs with Lupus
While SLE itself cannot be prevented in a predisposed dog, the frequency and severity of complications can be reduced with structured management:
- Minimise UV exposure – keep dogs with known photosensitivity indoors during peak sun hours, particularly in India’s climate; consider protective clothing for dogs with significant nasal or facial depigmentation
- Maintain a low-stress environment – predictable routines, reduced stressors, and appropriate mental engagement support immune stability
- Balanced nutrition – a complete, veterinarian-reviewed diet supports immune function and general health; in dogs with renal involvement, dietary protein levels are specifically managed
- Prompt treatment of infections – infections can trigger flares; any fever or sign of secondary infection in a dog with SLE should be assessed and treated early
- Consistent medication compliance – abrupt discontinuation of immunosuppressants is a common cause of relapse; owners should never adjust doses without veterinary guidance
Related Products
When to Seek Veterinary Care
Contact your veterinarian promptly if your dog with known SLE develops sudden weakness or inability to walk, a high or persistent fever, visible joint swelling, pale gums, skin ulceration that is worsening, significantly increased thirst and urination, or any acute change in their usual behaviour or activity level. Any of these signs in a dog already on immunosuppressive therapy may indicate a flare, an infection, or a drug side effect – all of which require assessment before the situation escalates.
Conclusion
SLE is one of the more demanding diagnoses in companion animal medicine, for the veterinarian working to confirm it, and for the pet parent managing it day to day. But it is not a hopeless diagnosis. Many dogs with SLE achieve stable, comfortable lives on appropriate treatment, with their disease well enough controlled that quality of life is maintained. What that requires is accurate diagnosis, a committed treatment plan, regular monitoring, and a pet parent who knows what to watch for. With those pieces in place, SLE becomes a condition that is lived with rather than defined by.